Specifications
- Product catalog number AB35SX040040080
-
Product catalog number AB35SX040040080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 40 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040060080
-
Product catalog number AB35SX040060080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 60 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040080080
-
Product catalog number AB35SX040080080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 80 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040100080
-
Product catalog number AB35SX040100080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 100 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040120080
-
Product catalog number AB35SX040120080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 120 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX050040080
-
Product catalog number AB35SX050040080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 40 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050060080
-
Product catalog number AB35SX050060080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 60 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050080080
-
Product catalog number AB35SX050080080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 80 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050100080
-
Product catalog number AB35SX050100080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 100 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050120080
-
Product catalog number AB35SX050120080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 120 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 16 atm
-
- Product catalog number AB35SX060040080
-
Product catalog number AB35SX060040080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 40 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060060080
-
Product catalog number AB35SX060060080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 60 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060080080
-
Product catalog number AB35SX060080080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 80 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060100080
-
Product catalog number AB35SX060100080 Sheath Size - 6F
Balloon Diameter - 6 mm
Balloon Length - 100 mm
Shaft Length - 80 cm
Nominal Pressure - 8 atm
Rated Burst Pressure - 14 atm
-
- Product catalog number AB35SX060120080
-
Product catalog number AB35SX060120080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 120 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 12 atm
-
- Product catalog number AB35SX040040135
-
Product catalog number AB35SX040040135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 40 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040060135
-
Product catalog number AB35SX040060135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 60 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040080135
-
Product catalog number AB35SX040080135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 80 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040100135
-
Product catalog number AB35SX040100135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 100 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040120135
-
Product catalog number AB35SX040120135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 120 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX050040135
-
Product catalog number AB35SX050040135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 40 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050060135
-
Product catalog number AB35SX050060135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 60 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050080135
-
Product catalog number AB35SX050080135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 80 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050100135
-
Product catalog number AB35SX050100135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 100 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050120135
-
Product catalog number AB35SX050120135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 120 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 16 atm
-
- Product catalog number AB35SX060040135
-
Product catalog number AB35SX060040135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 40 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060060135
-
Product catalog number AB35SX060060135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 60 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060080135
-
Product catalog number AB35SX060080135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 80 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060100135
-
Product catalog number AB35SX060100135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 100 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060120135
-
Product catalog number AB35SX060120135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 120 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 12 atm
-
- Product catalog number AB35SX040040080
-
Product catalog number AB35SX040040080 Sheath size - 6F
Balloon diameter - 4 mm
-
- Product catalog number AB35SX040060080
-
Product catalog number AB35SX040060080 Sheath size - 6F
Balloon diameter - 4 mm
-
- Product catalog number AB35SX040040080
-
Product catalog number AB35SX040040080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 40 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040060080
-
Product catalog number AB35SX040060080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 60 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040080080
-
Product catalog number AB35SX040080080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 80 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040100080
-
Product catalog number AB35SX040100080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 100 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040120080
-
Product catalog number AB35SX040120080 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 120 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX050040080
-
Product catalog number AB35SX050040080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 40 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050060080
-
Product catalog number AB35SX050060080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 60 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050080080
-
Product catalog number AB35SX050080080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 80 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050100080
-
Product catalog number AB35SX050100080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 100 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050120080
-
Product catalog number AB35SX050120080 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 120 mm
Shaft length - 80 cm
Nominal pressure - 10 atm
Rated burst pressure - 16 atm
-
- Product catalog number AB35SX060040080
-
Product catalog number AB35SX060040080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 40 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060060080
-
Product catalog number AB35SX060060080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 60 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060080080
-
Product catalog number AB35SX060080080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 80 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060100080
-
Product catalog number AB35SX060100080 Sheath Size - 6F
Balloon Diameter - 6 mm
Balloon Length - 100 mm
Shaft Length - 80 cm
Nominal Pressure - 8 atm
Rated Burst Pressure - 14 atm
-
- Product catalog number AB35SX060120080
-
Product catalog number AB35SX060120080 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 120 mm
Shaft length - 80 cm
Nominal pressure - 8 atm
Rated burst pressure - 12 atm
-
- Product catalog number AB35SX040040135
-
Product catalog number AB35SX040040135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 40 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040060135
-
Product catalog number AB35SX040060135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 60 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040080135
-
Product catalog number AB35SX040080135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 80 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040100135
-
Product catalog number AB35SX040100135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 100 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX040120135
-
Product catalog number AB35SX040120135 Sheath size - 6F
Balloon diameter - 4 mm
Balloon length - 120 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 20 atm
-
- Product catalog number AB35SX050040135
-
Product catalog number AB35SX050040135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 40 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050060135
-
Product catalog number AB35SX050060135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 60 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050080135
-
Product catalog number AB35SX050080135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 80 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050100135
-
Product catalog number AB35SX050100135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 100 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 18 atm
-
- Product catalog number AB35SX050120135
-
Product catalog number AB35SX050120135 Sheath size - 6F
Balloon diameter - 5 mm
Balloon length - 120 mm
Shaft length - 135 cm
Nominal pressure - 10 atm
Rated burst pressure - 16 atm
-
- Product catalog number AB35SX060040135
-
Product catalog number AB35SX060040135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 40 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060060135
-
Product catalog number AB35SX060060135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 60 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060080135
-
Product catalog number AB35SX060080135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 80 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060100135
-
Product catalog number AB35SX060100135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 100 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 14 atm
-
- Product catalog number AB35SX060120135
-
Product catalog number AB35SX060120135 Sheath size - 6F
Balloon diameter - 6 mm
Balloon length - 120 mm
Shaft length - 135 cm
Nominal pressure - 8 atm
Rated burst pressure - 12 atm
-
- 1. Lyden S, ILLUMENATE trial update, oral presentation, AMP Aug 2019.. Granada JF. Future directions, clinical applications and local drug delivery technologies. Presented at the Transcatheter Cardiovascular Therapeutics (TCT) 25th Annual Scientific Symposium; October 27-November 1, 2013; San Francisco, California. Slide 7, 15.
- 2. Mathews SJ, Stellarex in the Treatment of the SFA and Popliteal: Late- Breaking 3-Year Data, oral presentation, NCVH June 2019.
- 3. Lyden S, Safety And Effectiveness Of The Stellarex DCB With Low Dose Paclitaxel: 4 Year Results from the ILLUMENATE Pivotal trial, oral presentation, LINC Jan 2020, Leipzig, Germany.
- 4. Laird, et al. J Am Coll Cardio. 2015;66:2329-2338. In.Pact Summary of Safety and Effectiveness Data (SSED). Medtronic In.Pact Instructions for Use, M052624T001 Rev 1F.
- 5. Bard Lutonix Instructions for Use, BAW1387400r3.
- 6. Holden A. Comparing Trials Data in the Management of Calcified Arteries. Charing Cross 2018. April 24-26, 2018; London, UK.
- 7. Gray WA, Mortality assessment of ptx coated balloons - patient-level meta-analysis of the ILLUMENATE clinical program at 3 years, Circulation, 140:1145-1155, Oct 2019 1.
- 8. Lyden, S. LINC, 2019. Long-term safety data from the Stellarex DCB program Jan. 22, 2019. Leipzig, Germany.
- 9. Lyden S, Safety And Effectiveness Of The Stellarex DCB With Low Dose Paclitaxel: 4 Year Results from the ILLUMENATE Pivotal trial, oral presentation, LINC Jan 2020, Leipzig, Germany.
- 10. Diamantopoulos A, Gupta Y, Zayed H, KatsanosK. Paclitaxel-coated balloons and aneurysm formation in peripheral vessels. J VascSurg. 2015 Nov;62(5):1320-2.
- 11. Schmidt A et al. First experience with drug-eluting balloons in infrapoplitealarteries: restenosis rate and clinical outcome. J Am Coll Cardiol. 2011 Sep 6;58(11):1105-9.
- 12. LiistroF et al. Drug-eluting balloon in peripheral intervention for below the knee angioplasty evaluation (DEBATE-BTK): a randomized trial in diabetic patients with critical limb ischemia. Circulation. 2013 Aug 6;128(6):615-21.
- 13. Zeller T, et al. IN.PACT DEEP Trial Investigators. Drug-eluting balloon versus standard balloon angioplasty for infrapoplitealarterial revascularization in critical limb ischemia: 12-month results from the IN.PACT DEEP randomized trial. J Am Coll Cardiol. 2014 Oct 14;64(15):1568-76.
- 14. Venkatasubbu GD, et al. Surface modification and paclitaxel drug delivery of folic acid modified polyethylene glycol functionalized hydroxyapatite nanoparticles. Powder Technology. 2013;235:437-442.
- 15. Granada JF. Future directions, clinical applications and local drug delivery technologies. Presented at the Transcatheter Cardiovascular Therapeutics (TCT) 25th Annual Scientific Symposium; October 27-November 1, 2013; San Francisco, California. Slide 7, 15.
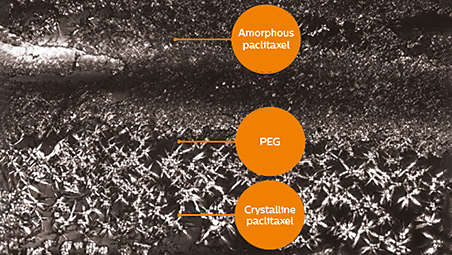
- 16. Stellarex: Data on file. Spectranetics Document. 2014. Spectranetics Pre-clinical Animal Study ADO097.
- 17. Gray B, Safety And Efficacy Of The Stellarex DCB With Low Dose Paclitaxel - 4 Year Results from the ILLUMENATE EU RCT, Viva Late Breaking Clinical Trials, 25 June 2020.
- *Complex patients refers to high rates of severe calcium, diabetes and renal insufficiency. Primary patency based on Kaplan-Meier estimates.
- †† No statistically significant difference in mortality
- ‡Data from independent CEC (clinical events committee) adjudication of all events resulting in death across all studies Product availability is subject to country regulatory clearance. Please contact your local sales representative to check the availability in your country.